Sharing economy service use is down, could be slow to recover after coronavirus
Washington, DC, April 6, 2020 -- A new Newsy/Ipsos poll finds that over half of sharing economy (such as ride sharing, car sharing, and clothing rental) consumers have used these services less in the past two weeks than they normally would. Once coronavirus-related quarantine guidelines are lifted, a larger number say they will use these services less, not more, than they did before.
Detailed Findings
- Once the coronavirus outbreak recedes, a quarter of sharing economy users plan to utilize theses services less than before.
- Forty percent of Americans report using services like ride sharing or clothing rental. More than half (54%) are currently using them less than they normally would.
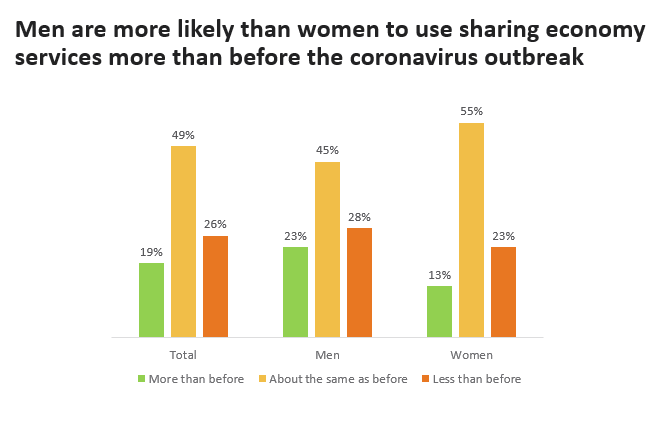
- Among these users, 26% plan to use them less than they did before the coronavirus outbreak, compared to 19% who plan to use them more. Half (49%) do not anticipate a change in their usage.
- Men and women differ on their plans for sharing economy services. Men are more likely than women say they will use these services more than before the COVID-19 pandemic.
About the Study
These are some of the findings of an Ipsos poll conducted between April 1-2, 2020, on behalf of Newsy. For this survey, a sample of 1,005 adults age 18+ from the continental U.S., Alaska, and Hawaii was interviewed online in English.
The sample for this study was randomly drawn from Ipsos’ online panel (see link below for more info on “Access Panels and Recruitment”), partner online panel sources, and “river” sampling (see link below for more info on the Ipsos “Ampario Overview” sample method) and does not rely on a population frame in the traditional sense. Ipsos uses fixed sample targets, unique to each study, in drawing a sample. After a sample has been obtained from the Ipsos panel, Ipsos calibrates respondent characteristics to be representative of the U.S. Population using standard procedures such as raking-ratio adjustments. The source of these population targets is U.S. Census 2016 American Community Survey data. The sample drawn for this study reflects fixed sample targets on demographics. Posthoc weights were made to the population characteristics on gender, age, race/ethnicity, region, and education.
Statistical margins of error are not applicable to online non-probability polls. All sample surveys and polls may be subject to other sources of error, including, but not limited to coverage error and measurement error. Where figures do not sum to 100, this is due to the effects of rounding. The precision of Ipsos online polls is measured using a credibility interval. In this case, the poll has a credibility interval of plus or minus 3.5 percentage points for all respondents. Ipsos calculates a design effect (DEFF) for each study based on the variation of the weights, following the formula of Kish (1965). This study had a credibility interval adjusted for design effect of the following (n=1,005, DEFF=1.5, adjusted Confidence Interval=+/-5.0 percentage points).
For more information on this news release, please contact:
Mallory Newall
Director, US
Public Affairs
+1 202 420-2014
[email protected]
Kate Silverstein
Media Relations Specialist, US
Public Affairs
+1 718 755-8829
[email protected]
About Ipsos
Ipsos is the world’s third largest market research company, present in 90 markets and employing more than 18,000 people.
Our passionately curious research professionals, analysts and scientists have built unique multi-specialist capabilities that provide true understanding and powerful insights into the actions, opinions and motivations of citizens, consumers, patients, customers or employees. We serve more than 5000 clients across the world with 75 business solutions.
Founded in France in 1975, Ipsos is listed on the Euronext Paris since July 1st, 1999. The company is part of the SBF 120 and the Mid-60 index and is eligible for the Deferred Settlement Service (SRD).
ISIN code FR0000073298, Reuters ISOS.PA, Bloomberg IPS:FP www.ipsos.com



